Fact
ba
.
se

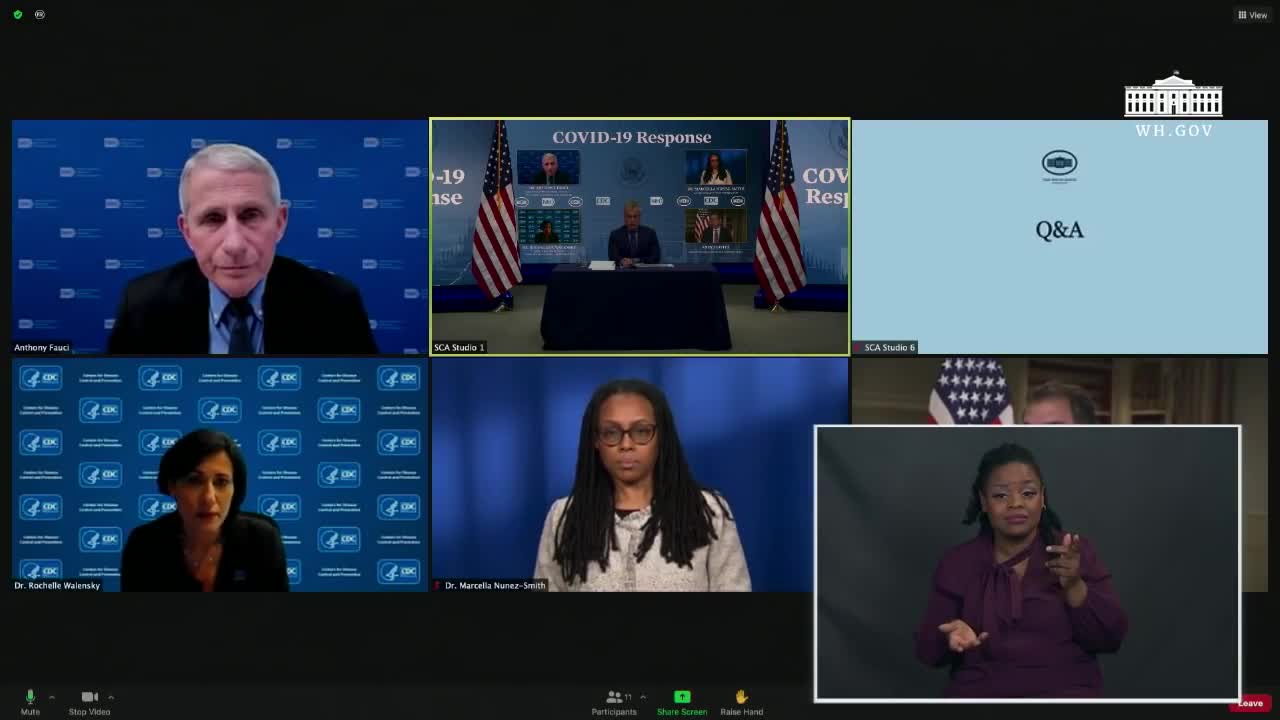
Press Conference: White House COVID-19 Response Team Holds a Briefing - January 27, 2021
-
Andy Slavitt Person
-
Rochelle Walensky Person
-
Jeff Zients Person
-
Marcella Nunez-Smith Person
-
Anthony Fauci Person
-
Question Person
-
Operator Person
Jeff Zients
Very Positive

00:00:00-00:00:28 (28 sec)

"Okay. Thank you, everyone, for joining us today. I am Jeff Zients, White House COVID coordinator. I have the privilege of being joined by four of America's leading doctors and health experts today to conduct our first COVID-19 briefing. I'll keep my comments very brief. Yesterday, we met with governors and other local leaders to update them on our progress and plans."
1
Jeff Zients
Very Positive

00:00:28-00:00:57 (29 sec)

"Today we are here because we have to have a regular series -- we want to have a regular series of briefings to engage you on the state of the pandemic, engage the American people and all of you, to ensure everyone has accurate and up-to-date information. The president strongly believes that the scientists who are leading the effort should communicate directly to the American people."
2
Jeff Zients
Somewhat Positive

00:00:57-00:01:29 (32 sec)

"To that end, going forward you can expect to hear directly from people, including Dr. Fauci, Dr. Walensky, Dr. Nunez-Smith, and Surgeon General Dr. Murphy. My role as coordinator of our federal response is to ensure that we are empowering our experts and using the full capabilities of the U.S. government to respond to and recover from this pandemic."
3
Jeff Zients
Very Positive

00:01:29-00:01:55 (26 sec)
"I will participate in these briefings periodically and have asked Andy Slavitt, Senior Advisor to our White House COVID-19 response, to lead [Inaudible]. The president believes the federal government should be the source of truth for the public to get clear, accessible, and scientifically accurate information about the pandemic."
4
Jeff Zients
Positive

00:01:55-00:02:20 (25 sec)

"We will not always know the answers to your questions. When we don't, we will tell you. We will update you on our progress and we will be open about any setbacks along the way. We will always be transparent and straightforward with all of you. During these briefings, you'll generally hear about three topics."
5
Jeff Zients
Very Positive

00:02:20-00:03:08 (48 sec)

"First, the current state of the pandemic, including data on testing, infections, hospitalizations, and deaths. Second, developments on the scientific front, including in areas around vaccines, therapeutics, and possible variants. And third, operational updates and how we're executing against the president's national strategy, including our efforts to get shots in arms, increase testing, secure adequate supplies of PPE and other critical materials, and reopen K-8 schools."
6
Jeff Zients
Very Positive

00:03:08-00:03:46 (37 sec)
"And we will provide regular updates on our efforts to ensure that equity remains at the core of all we do, that those who need [Inaudible] our strategy and execution. I want to highlight an important action that we'll take today to expand the number of vaccinators in the field to put needles in arms. HHS will amend the current Public Readiness and Emergency Preparedness Act, otherwise known as the PREP Act."
7
Jeff Zients
Very Positive

00:03:46-00:04:09 (23 sec)

"They're going to amend it to permit retired doctors and nurses to administer shots, and also to permit licensed doctors, nurses, and other practice -- practitioners who are licensed in their home states to administer shots across state lines. As the president said, we need to increase the number of places where people can get vaccinated, and also at the same time increase the number of vaccinators."
8
Jeff Zients
Very Positive

00:04:09-00:04:18 (9 sec)
"This action by HHS today will help get more vaccinators in the field. We're lucky to have so many trusted experts on our team to help the country fight and recover from this pandemic, and everyone will benefit from hearing directly from them. So with that, I'll turn it over to Dr. Walensky at the CDC. Dr. Walensky."
9
Rochelle Walensky
Neutral

00:04:18-00:04:18 ( sec)

"[Inaudible]"
10
Jeff Zients
Slightly Negative

00:04:18-00:04:25 (7 sec)
"Dr. Walensky, can you hear us? I think we're having some technical difficulties. Dr. Fauci."
11
Anthony Fauci
Neutral

00:04:25-00:04:25 ( sec)

"[Inaudible]"
12
Jeff Zients
Slightly Positive

00:04:25-00:04:27 (2 sec)

"Okay. Dr. Walensky, can you hear us?"
13
Rochelle Walensky
Neutral

00:04:27-00:04:30 (2 sec)

"I can. Can you hear me?"
14
Jeff Zients
Positive

00:04:30-00:04:33 (4 sec)
"Great. We had some operational difficulties. We're over to you on the state of the pandemic. Dr. Walensky from the CDC."
15
Rochelle Walensky
Very Positive

00:04:33-00:06:12 (98 sec)

"Thank you, Jeff, and thank you all for joining us today. My name is Dr. Rochelle Walensky, and I am the new director of the CDC. I'd like to start by recognizing the truly tragic toll that the COVID-19 pandemic has taken on all of us, especially those who have lost loved ones and who continue to suffer in the wake of this disease."
16
Rochelle Walensky
Very Positive

00:06:12-00:06:43 (32 sec)
"This is the first of many briefings and I look forward to speaking with you often, updating you on the state of the pandemic, and sharing continued actions that the public can take to protect themselves and their loved ones. Let's start with an overview of the data. There are some hopeful signs that we are watching very closely. 25.1 million COVID-19 cases have been reported to the CDC through January 25th."
17
Rochelle Walensky
Neutral

00:06:43-00:07:08 (24 sec)

"During the week of January 19th through the 25th, the seven-day average of new cases decreased by 21 percent to over 160,000 -- 66,000 per day. During the week of January 18th to 24th, the seven-day average of new hospital admissions of patients with COVID-19 also decreased by 15 percent, to nearly 13,000 per day."
18
Rochelle Walensky
Very Positive
00:07:08-00:07:57 (49 sec)

"However, over 78,000 patients were hospitalized with COVID-19 as of January 24th. 419,827 deaths have been reported since January 22, 2020. During the week of January 19th to January 25th, the seven-day average number of deaths decreased by 4.9 percent to 3,113 per day. And while these data are trending downward is a good sign, the number of deaths reported in a single day during the outbreak was reported on January 20th, the peak number of 4,383 deaths."
19
Rochelle Walensky
Somewhat Positive
00:07:57-00:08:25 (29 sec)
"Though I am encouraged by these trends, our case rates remain extraordinarily high, and now is the time to remain vigilant. If we continue on the current trajectory, the CDC most recent national ensemble forecasts, predicts, that 479,000 to 514,000 COVID-19 deaths will be reported by February 20, 2021. I know this is not news we all want to hear, but this is something we must say so we are all aware."
20
Rochelle Walensky
Very Positive
00:08:25-00:08:59 (34 sec)
"But if we are united in action, we can turn things around. Continuing to expand safe, effective vaccination is key to ending the COVID-19 pandemic and bringing our country back to health. Daily vaccine doses administered continued to rise to more than 1.6 million doses per day over the past week. Over 23.5 million doses of COVID-19 vaccine have been already administered."
21
Rochelle Walensky
Very Positive
00:08:59-00:09:30 (31 sec)
"This includes 3.4 million people who have already received their second dose. I want to take a moment here and emphasize that the COVID-19 vaccines are safe, and they work. And this is backed up with data that I would like to share with you. Last week the CDC released an MMWR on severe adverse events, most notably anaphylaxis, for those receipt of the Moderna vaccine as well as the Pfizer vaccine."
22
Rochelle Walensky
Very Positive
00:09:30-00:09:54 (24 sec)
"Based on our most recent data, we found that there were 2.1 cases of anaphylaxis per million administered doses of Moderna, and 6.2 cases of anaphylaxis per million doses administered of Pfizer. Let me be clear, these are rare, treatable outcomes, and the COVID-19 vaccines are safe. It's also important to put this into context."
23
Rochelle Walensky
Very Negative
00:09:54-00:10:17 (22 sec)
"The risks of getting sick with COVID-19 are much higher than the risks of allergy or anaphylaxis from the vaccine. In the U.S., sadly, approximately 16,500 people for every 1 million who are diagnosed with COVID will die. And while anaphylaxis can be scary, there are effective treatments, and patients generally do quite well."
24
Rochelle Walensky
Slightly Negative
00:10:17-00:10:51 (34 sec)
"It's also important to note the mild side effects, like pain where you got your shot, feeling feverish or tired, and muscle aches after getting your shot. These are all normal and expected part of getting the vaccine, especially the second dose. And these symptoms mean that your immune system is revving up and the vaccine is actually working."
25
Rochelle Walensky
Very Positive
00:10:51-00:11:13 (22 sec)
"CDC is actively monitoring for safety signals associated with these vaccines, and we are committed to sharing those safety data with you as they emerge. Also last week, CDC updated guidance to reduce barriers associated with returning for a second vaccine dose on a specific date or when circumstances change between a first and second dose."
26
Rochelle Walensky
Very Positive
00:11:13-00:11:39 (26 sec)
"There are two important updates that I want to clarify. First, in our updated guidance the CDC still recommends that people get their second dose as close to the recommended interval as possible. That would be three weeks for the Pfizer vaccine and four weeks for the Moderna vaccine. However, we also know that life can get in the way, and that some of those doses may be missed in the best time window."
27
Rochelle Walensky
Positive
00:11:39-00:12:03 (24 sec)
"And in those rare circumstances, the second dose may be given up to six weeks or 42 days after the first. Second, the CDC has reiterated that the COVID-19 vaccines are not interchangeable. However, it's also true than in extremely rare circumstances, people may not simply remember or have documentation of which first dose they received."
28
Rochelle Walensky
Slightly Negative
00:12:03-00:12:28 (25 sec)

"In these extremely rare situations, we have said that any available mRNA COVID-19 vaccine may be administered as the second dose if people are unaware of which first dose they received, and that would be at least 28 days after their first. I also know that there are concerns about variants. Viruses mutate, and we have always expected that variants would emerge and we have been looking for them."
29
Rochelle Walensky
Very Positive

00:12:28-00:12:53 (25 sec)
"The variants that have been identified recently seem to spread more easily, they're more transmissible, which can lead to increased number of cases and increased stress on our already taxed healthcare system. In the United States, 308 cases of B117 variants that originated from the UK have been confirmed in 26 states as of January 26th."
30
Rochelle Walensky
Very Positive
00:12:53-00:13:19 (26 sec)

"We also identified this week our first case of the P1 variant in the United States in Minnesota. To date, no cases of the B1351 variant that was first detected in South Africa has been identified in the U.S. CDC is committed to working with international and state and local partners, and increasing surveillance to monitor the situation and share as soon as we learn more."
31
Rochelle Walensky
Very Positive
00:13:19-00:13:42 (23 sec)
"The emergency -- the emergence of variants underscores the need for public health action. First, get vaccinated when it's your turn. Also, some people may need help getting vaccinated. Please consider helping your neighbors and loved ones schedule or travel to their appointments. Second, wear a mask, practice social distancing, and wash her hands."
32
Rochelle Walensky
Very Positive
00:13:42-00:13:57 (15 sec)
"And finally, and now is not the time to travel. But if you must, be safe and follow the CDC guidance. This includes wearing a mask, as well as testing and quarantine protocol. Please do your part to get cases down by simply taking these actions. Thank you. I will now turn it over to Dr. Fauci who will share some scientific updates."
33
Anthony Fauci
Neutral
00:13:57-00:14:00 (3 sec)

"[Inaudible] -- a bit about therapeutics. In therapeutics --"
34
Jeff Zients
Neutral
00:14:00-00:14:01 ( sec)

"-- Dr. Fauci?"
35
Anthony Fauci
Slightly Positive

00:14:01-00:14:01 ( sec)

"Yeah?"
36
Jeff Zients
Very Positive
00:14:01-00:14:05 (3 sec)
"I believe your audio just came on. So if you could come back to the beginning and start again that'd be great."
37
Anthony Fauci
Very Positive
00:14:05-00:15:15 (70 sec)

"Okay. So in other words nobody heard anything from me. Is that correct? Okay. Well I said a lot of really good stuff. Just let -- thank you, Dr. Walensky. What I'm gonna do is talk about three areas. One is therapeutics, the other is a summary of where we are with vaccines, and the other is looking at some of the implificat -- implications of the variants of concern that Dr. Walensky mentioned."
38
Anthony Fauci
Slightly Positive
00:15:15-00:15:36 (22 sec)
"When you think in terms of therapeutics you think in terms of therapeutics for advanced disease and therapeutics for early and moderate disease. But also buried in that is targeting the virus itself versus targeting the aberrant inflammatory responses that often are the source and the cause of the morbidities and mortalities that we see with this particular disease."
39
Anthony Fauci
Very Positive

00:15:36-00:15:57 (21 sec)
"So first, if you look at the therapeutics for moderate advanced disease, one of the ones that are shown clearly to be very efficacious is Dexamethasone in people who are hospitalized who are on ventilators or have high flow oxygen requirements. It is shown to significantly diminish the 28-day mortality."
40
Anthony Fauci
Slightly Positive

00:15:57-00:16:20 (22 sec)

"There have been -- been another -- a group of studies such as Pyrazinamide and Remdesivir which have received an Emergency Use Authorization. Moving over to the left-hand part of the slide, there are a number of interventions that have been used for more early or moderate disease. For example, Remdesivir, which is a direct antiviral, has received FDA approval."
41
Anthony Fauci
Very Positive

00:16:20-00:16:50 (30 sec)

"And a variety of monoclonal antibodies have also received Emergency Use Authorization as have convalescent plasma. Other antivirals, hyperimmune globulin, and things like anticoagulants are currently in clinical trial. One comment about things like monoclonal antibodies and convalescent plasma. Since they are giving antibody to someone who would need a direct antiviral effect of the antibody it is best to use it early, before individuals actually have had the opportunity to progress."
42
Anthony Fauci
Positive
00:16:50-00:17:20 (30 sec)

"And that's what we're gonna be seeing in the future is an attempt to as early as possible get these interventions to the patients in their particular state. Hopefully as outpatients if we can logistically get that done. If we could move on to the next slide, and that is vaccines. As many of you know now, the federal government has been involved either in the development of and or the facilitation of the testing of a number of vaccines that represent three separate platforms."
43
Anthony Fauci
Very Positive
00:17:20-00:17:49 (30 sec)

"Shown on this slide is what we all know is the extraordinary, efficacious, and safe results of the Moderna and the Pfizer-BioNTech product with 94 to 95 percent efficacy and a good safety profile, both having received an EUA. What we're looking at closely now is others that you see on the slide, Johnson, Astra-Zeneca, and Novavax which are in various states of trial."
44
Anthony Fauci
Very Positive

00:17:49-00:18:13 (23 sec)
"Of particular importance that you'll be hearing about likely within the next few days to a week are the results from the Johnson J and J trial. And the reason they're important, it relates to some of the things that Dr. Walensky said. And that is it's a phase three trial with a different platform, a human adenovirus vector."
45
Anthony Fauci
Very Positive

00:18:13-00:18:40 (27 sec)
"But it is going to be looking at efficacy not only in the United States but also in South Africa and in Brazil. So we will get a good feel from those, I hope. I believe we will. We'll let the data speak for itself. But what we will see was the relative efficacy against the wild type virus that is predominantly in the United States as well as the South African isolate which in fact is something that we will be able to get a feel for the efficacy there."
46
Anthony Fauci
Very Positive

00:18:40-00:19:05 (25 sec)
"So we'll have some comparative efficacy which will inform us on where we would go if the eventuation occurs that we do have that particular lineage that would take a sense -- seed itself in the United States. So let me move on to the last slide because that relates to what I said. Dr. Walensky gave you a good feel for the distribution of the various lineages."
47
Anthony Fauci
Very Positive
00:19:05-00:19:43 (38 sec)

"The 117 which is in the U.K., the 351 South Africa which I just mentioned, and the P1 in Brazil. She mentioned about the increased transmissibility and the likelihood of an increase actually in some lethality in the B117. The important question that people ask is what is the impact on both monoclonal antibodies and on the vaccine induced antibodies related to the vaccines that we are currently distributing now."
48
Anthony Fauci
Negative
00:19:43-00:20:15 (33 sec)

"When you're looking at the 117 namely what we refer to as the U.K. variant, what we're seeing is a very slight if at all impact on vaccine induced antibodies and very little impact on anything else. So we are covered with that. Things get a bit more problematic when you go to the 351, or what we're seeing at the dominant one in South Africa."
49
Anthony Fauci
Very Positive
00:20:15-00:20:40 (24 sec)
"Because in that regard there is a moderate diminution, namely a multifold diminution in the in-vitro neutralization by vaccine induced antibodies. However, and this is an important however, it still is well within the cushion of protection. So you could diminish the vaccine induced antibody efficacy by a few fold and still be well within the protective range of the vaccine."
50
Anthony Fauci
Neutral

00:20:40-00:21:07 (28 sec)
"And that's the reason why you've seen announcements that actually the vaccines that we're using are still effective. However, given that as a -- a fact now we have to be concerned looking forward at what the further evolution of this might be. Now, I wanna make one mention about the monoclonal antibodies because they are more seriously inhibited in the sense of impacting on their efficacy by this South African strain."
51
Anthony Fauci
Neutral
00:21:07-00:21:29 (22 sec)

"That's the reason why there will be attempts to develop even other antibodies that might be able to avert this particular problem. So looking forward what we plan to do in collaboration with the companies is to develop what we would call alternative or boosts that would use the same platform but that would incorporate a particular immunogen that would address these particular variants."
52
Anthony Fauci
Leans Negative
00:21:29-00:21:40 (10 sec)
"And for that reason we will always want to be a step or two ahead of what might be a problem in the future. So I'll stop there now and hand it over to Andy Slavitt."
53
Andy Slavitt
Very Positive

00:21:40-00:22:06 (26 sec)
"Thank you, Dr. Fauci. My audio on? Yep. Thank you, Dr. Fauci. And also thank you Dr. Walensky. The country is truly lucky to have the two of you leading the charge on both our nation's best scientists and our nation's best public health officials. I wanna pick up where Drs. Walensky and Fauci left off, public health and science."
54
Andy Slavitt
Slightly Negative
00:22:06-00:22:32 (26 sec)

"I'm gonna talk about another topic, which is execution of the plan to combat the COVID crisis. Namely, the strategy, the progress, and the challenges openly and transparently. Now to begin, I'd like to start with one of the core pillars of our plan, and that is the equitable healthcare access and outcomes for all Americans."
55
Andy Slavitt
Positive

00:22:32-00:22:48 (17 sec)

"So, let me introduce Dr. Marcella Nunez-Smith, who is head of the Health Equity Task Force to make some comments on where we stand as a country in the battle to make sure we get every American equitable access to medical care and the vaccinations that they need."
56
Marcella Nunez-Smith
Very Positive

00:22:48-00:23:11 (23 sec)
"Great. Thank you so much Andy. And that's correct. You know, equity is absolutely a foundational component of our national plan. You know -- there's a moral imperative. It's simply the right thing to do to ensure that those who are hardest hit or at highest risk have the opportunity to stay safe and healthy through this pandemic."
57
Marcella Nunez-Smith
Very Negative

00:23:11-00:23:37 (25 sec)
"And there's also the science of it. We cannot beat this virus without making sure we're executing a plan that works for all communities. So we've committed to taking an equity lens to every aspect of this pandemic response. That includes minimizing the risk of getting infected and for those who get COVID minimizing the risk of hospitalization or death."
58
Marcella Nunez-Smith
Somewhat Negative
00:23:37-00:24:14 (38 sec)

"So that includes insuring access to PPE, testing, treatments, and vaccines. It also includes making it possible for people to safely quarantine and isolate and to monitor the impacts of long COVID. So from what the data show us, we still have a long way to go. Latino, indigenous, and black people in America are dying at high rates from COVID-19. And folks in rural communities have case and death rates that continue to climb."
59
Marcella Nunez-Smith
Very Negative

00:24:14-00:24:59 (44 sec)
"And they're dying at higher rates than people in metropolitan areas. So by race, ethnicity, sexual orientation, gender identity -- you know -- disability, geography -- you know -- based on where you live, COVID-19 is leaving a terrible imprint on far too many communities. Next slide. So the president took immediate action to combat COVID-19 health equities."
60
Marcella Nunez-Smith
Very Positive

00:24:59-00:25:26 (28 sec)
"Last Thursday, President Biden signed an executive order on insuring an equitable pandemic response and recovery. And a key part of his approach to ensuring equity is through the COVID-19 Health Equity Task Force. So this task force is responsible for providing specific recommendations to the president through the COVID-19 response coordinator for mitigating the health inequities caused or exacerbated by the COVID-19 pandemic."
61
Marcella Nunez-Smith
Positive

00:25:26-00:25:50 (24 sec)

"And also for preventing these kinds of inequities in the future. You know -- the task force will include individuals with insights into these groups that have been hardest hit. So their role will be advisory in nature but the president has asked them to make some recommendations on a range of issues. Optimally allocating COVID-19 resources by agencies and by state, local, tribal, and territorial officials."
62
Marcella Nunez-Smith
Very Positive

00:25:50-00:26:19 (29 sec)
"Dispersing COVID-19 relief funds in a way that advances equity. You know -- ensuring effective, culturally aligned communication, messaging, and outreach to our hardest hit communities. And addressing ongoing health inequities faced by COVID-19 survivors that may merit a public health response. So it's gonna be essential that we collect data for the hardest hit communities and identify data sources that would enable development of short-term targets for pandemic related actions."
63
Marcella Nunez-Smith
Leans Positive

00:26:19-00:26:39 (20 sec)

"And address longer term data shortfalls and challenges to better prepare and respond to future pandemics. So we will be announcing the members of this task force soon, as President Biden is adamant they hit the ground running, especially now as so many people and communities are disproportionately hurting from this pandemic."
64
Marcella Nunez-Smith
Very Positive

00:26:39-00:27:06 (27 sec)
"And for the last slide. You know -- finally, it's critical everyone has equitable access to all of the resources necessary to make it through this pandemic. And that includes equitable distribution of COVID-19 vaccines to all communities. So all Americans, everyone in our country should have the benefit of a safe, effective vaccine that can prevent them from getting sick or dying from COVID-19. It takes intention and deliberate action to advance equity in this vaccine distribution process."
65
Marcella Nunez-Smith
Somewhat Positive

00:27:06-00:27:24 (19 sec)
"So we're gonna do that through a series of important actions. We're leveraging data sources both from government and other sectors to find and remedy inequities. You know -- we're limiting all out of pocket costs for vaccines. Your ability to pay should not play a role in your decision of whether or not to receive this vaccine."
66
Marcella Nunez-Smith
Very Positive
00:27:24-00:27:48 (23 sec)

"We're removing structural barriers to access in underserved communities. We'll do this by making sure that it's convenient and accessible to get to vaccination sites, by increasing the clinical and community-based workforce for outreach, education, vaccination, and wrap around services. You know -- we're working to make sure that transportation and paid time off are available so people can make it out to get their vaccine."
67
Marcella Nunez-Smith
Very Positive

00:27:48-00:28:03 (16 sec)
"We're gonna increase and support core venues of vaccination including federally qualified health centers, and Andy's gonna talk about some of our other venues for vaccination that we're ramping up. But it will be important to make sure vaccine is available for everyone and oftentimes that will mean bringing the vaccine right to people."
68
Marcella Nunez-Smith
Very Positive

00:28:03-00:28:28 (24 sec)
"So to -- next we're already talking to states about their pandemic plans and strategies for equity. We're here to provide technical assistance when needed, but when local health officials are making great progress in equity we're sharing some of those great ideas with their peers around the country. And finally, we're launching a robust national public education campaign to make sure people know about this vaccine, the facts and not the misinformation."
69
Marcella Nunez-Smith
Very Positive

00:28:28-00:28:44 (17 sec)
"We want people to know that the vaccines are safe and effective. We want everyone to be able to make decisions with the best information possible, and we're gonna make sure that the information comes to them in places and in ways that are most likely to reach them. It's critical this public education work be very local in nature."
70
Marcella Nunez-Smith
Very Positive

00:28:44-00:29:03 (19 sec)
"Critical that we in the federal government support the relationships that already exist between faith and community leaders and their communities. So while there is no single way to do this work, there are best practices that must define our approach. And supporting those best practices will be our focuses in the days or weeks to come."
71
Marcella Nunez-Smith
Very Positive

00:29:03-00:29:17 (14 sec)

"So there is so much to do to get through this pandemic and we want to make sure all communities are cared for and supported along the way. So with that, I'm going to turn it back over to you, Andy."
72
Andy Slavitt
Very Positive

00:29:17-00:29:41 (24 sec)

"Thank you, Dr. Nunez-Smith. And look, it's appropriate that I follow in my report Drs. Walensky, Fauci, and Nunez-Smith very simply because it's important to send the message to the public that the White House respects and will follow the science and the scientists will speak independently and that our core values of science, public health, and equity are going to drive our actions here at the White House."
73
Andy Slavitt
Very Positive

00:29:41-00:30:02 (20 sec)
"The president launched a national strategy just as he assumed office on his second day. And at the heart of that plan is a most the most aggressive actions possible to stem the pandemic and follow the science. So Dr. Walensky spoke about the high levels of spread still in this country, even as they decline."
74
Andy Slavitt
Very Negative

00:30:02-00:30:26 (24 sec)

"Dr. Fauci talked about the threat from the new variance and Dr. Nunez-Smith spoke about our difficulty and challenges in getting access to vaccines all across the country. So given those realities, accelerating the availability in the administration of vaccines are all the more important. So it's been a busy seven days."
75
Andy Slavitt
Very Positive

00:30:26-00:31:04 (38 sec)
"You've seen us begin to execute our strategy. We've set up a team, began working with states in vaccine manufacturers, launched community vaccination centers, taken action to improve supply, revised travel restrictions, and more. Now, I know that many Americans are anxious and eager to get vaccinated. I want you to know we are taking this issue on with incredible urgency and purpose, despite not inheriting a fully developed strategy or the infrastructure to make vaccines readily available to Americans as quickly as they need to be."
76
Andy Slavitt
Somewhat Positive

00:31:04-00:31:25 (21 sec)
"So far this week, we've been hitting our target of an average of 1 million vaccinations per day necessary to meet the president's early commitment to administer 100 million shots in 100 days. Now, as you heard from the president this week, he is pushing us to view 1 million per day as the floor, not the ceiling."
77
Andy Slavitt
Very Positive

00:31:25-00:32:00 (35 sec)
"To that end, we are releasing more supply, activating FEMA, and deploying many more personnel. In fact, just this week, FEMA announced that they had obligated $1 billion to support state vaccination sites. Now, I want to step back for a moment and put our vaccination plan in some context. To vaccinate every American over the age of 16, we are looking at a total need of well over 500 million doses of vaccines."
78
Andy Slavitt
Somewhat Positive

00:32:00-00:32:29 (29 sec)

"Now, that assumes two shots for every American 16 and older. Now, I want to pause and say that everything we're talking about today in terms of vaccines reflects the vaccine is currently on the market. That is a two-dose regimen. We are not counting on things that are not in existence today. Our plan will be to make sure that we prepare for as many contingencies as exist."
79
Andy Slavitt
Positive

00:32:29-00:32:59 (30 sec)

"Right now, I want to level with the public that we are facing two constraining factors. The first is getting enough supply quickly enough, and the second is the ability to administer the vaccines quickly once they are produced and sent out to the sites. We are taking action to increase supply and increase capacity, but even so, it will be months before everyone who wants a vaccine will be able to get one."
80
Andy Slavitt
Very Positive

00:32:59-00:33:27 (27 sec)

"Now, let me start with what happened so far. We've delivered 47 million doses to states and long-term care facilities and we've administered about 24 million doses. Now, we've gotten several questions on this, so I wanted to be clear. As you heard us announce yesterday, any stockpile that may have existed previously no longer exists."
81
Andy Slavitt
Very Positive

00:33:27-00:33:56 (30 sec)
"Our practice is to maintain a rolling inventory of two to three days of supply that we can use to supplement any shortfalls in production and to ensure that we are making deliveries is committed. But we are passing doses directly along to states very much in real time as they order them, and we are confident that we will receive the supply on a rolling basis from the manufacturers to allow us to continue to do so."
82
Andy Slavitt
Very Positive

00:33:56-00:34:21 (24 sec)
"Now, we know there are places in the country with not enough vaccines and at the same time, there are places with vaccines that they are not using yet. This is a natural challenge states are facing and we will give them more visibility into their forthcoming supply and providing technical assistance that will help them make sure to make quicker use of these doses."
83
Andy Slavitt
Very Positive

00:34:21-00:34:49 (29 sec)
"But I want to emphasize an essential ingredient to increasing the administration of vaccines is passage of the American Rescue Plan, which dedicates needed money for vaccinators and for the administration of vaccines. Now, let me take it through what we were doing to increase the supply. So the president evoked invoked the Defense Production Act last week as a big first step to increase the supply of vaccines."
84
Andy Slavitt
Very Negative

00:34:49-00:35:21 (31 sec)
"We're going to make sure that we get six doses out of Pfizer's vials everywhere in America because that's the potential. And we're going to do that by acquiring something called low dead space syringes. I'm sure everybody knows what low dead space syringes are. I now do. And yesterday, we announced a 16 percent increase in supply flowing to states every week for the next 3 weeks to a minimum of 10 million doses per week."
85
Andy Slavitt
Very Positive

00:35:21-00:35:40 (19 sec)
"That's good news. But what's also important about it is it gives states the visibility that they have lacked to know how many vaccines are coming in so they can make a commitments to their partners in States and to the citizens and residents of their state so they know when to expect vaccines to arrive."
86
Andy Slavitt
Very Positive

00:35:40-00:36:19 (39 sec)
"Pfizer and Moderna are committed to delivering a total of 200 million doses by the end of March with much of it coming at the end of the quarter. So it will accelerate. Pfizer yesterday announced that they think they can deliver 120 million doses this quarter. And as you heard from the president yesterday, the United States plans to purchase an additional 200 million doses from Moderna and Pfizer this year, which will be sufficient to vaccinate every American over the age of 16 and we expect those doses this summer."
87
Andy Slavitt
Very Positive

00:36:19-00:36:43 (24 sec)
"This entire thing is an unprecedented undertaking. We will run into anticipate -- unanticipated issues, but our plan and the actions we are taking in the days and weeks ahead is to deliver on these commitments to the public. Finally, we've also announced that we are creating additional paths to get Americans vaccinated."
88
Andy Slavitt
Leans Negative

00:36:43-00:37:21 (38 sec)
"These will include standing up what we call community vaccination centers. We are going to launch 100 community vaccination centers this month. We will be also supplying more -- we will be supplying vaccines directly to the pharmacies, we will be standing up mobile clinics to reach hard to reach areas, particularly in -- for pursuit of the health equity goals that Dr. Nunez-Smith has outlined and we're going to partner with community health centers that reach hard hit communities and have infrastructure to deliver vaccines."
89
Andy Slavitt
Very Positive

00:37:21-00:37:44 (23 sec)
"But again, I want to close with this, it's critically important that Congress act. The American Rescue Plan is an important and vital part of getting America vaccinated. It's got bold and ambitious legislation that will help fund the effort that we're outlining today in providing the resources we need to defeat this pandemic."
90
Andy Slavitt
Neutral

00:37:44-00:37:59 (14 sec)

"So we need immediate action. With that, I'm going to pass it back so that we can take some of your questions."
91
Operator
Very Positive

00:37:59-00:38:12 (13 sec)

"Okay, great. If folks have a question, you can use the raise hand feature on your screen and we will try to get to as many as we can here in the time that we have left. So let's start with Angela Lee at Yahoo news. You should be unmuted."
92
Question
Somewhat Positive

00:38:12-00:38:32 (19 sec)

"Hi there. Thank you for taking questions. Can you hear me?"
93
Operator
Slightly Positive

00:38:32-00:38:33 (1 sec)

"Yeah, we can."
94
Question
Very Positive

00:38:33-00:38:46 (13 sec)
"All right, great. Thanks so much. I just wanted to get some clarity about the rollouts of these vaccines. It seems like we are getting an increase in vaccinations and administration. Can you clarify what the sudden change was to be able to get to this point?"
95
Jeff Zients
Neutral

00:38:46-00:38:57 (12 sec)
"Andy? Andy Slavitt, can you hear us?"
96
Andy Slavitt
Positive

00:38:57-00:39:00 (3 sec)

"Yes. I can hear you."
97
Jeff Zients
Very Positive

00:39:00-00:39:15 (15 sec)
"Okay, did you hear the question? It was about what increase, the recent increase and what it was driven by. The minimum of 10 million to states for the next three weeks, how is that achieved, Andy?"
98
Andy Slavitt
Very Positive

00:39:15-00:39:43 (28 sec)
"Thank you. Yeah, thank you, Jeff. That's a good question. Look, this -- this entire effort is not about just big blast, goals, but it's about day-to-day execution. You know, this is the result of the work of Pfizer and Moderna and, in this case, much of the increase came from our work with Moderna that we're -- that were planned increases and scrutinizing what's available so that we can make a commitment to states."
99
Andy Slavitt
Slightly Positive

00:39:43-00:39:51 (9 sec)
"Very importantly, we need to add predictability into this process. So I think this is due to the hard work of everybody involved in the process."
100
Operator
Very Positive

00:39:51-00:39:59 (8 sec)
"Great. Let's go to Cheryl Stolberg at the Times next."
101
Question
Very Positive

00:39:59-00:40:25 (25 sec)

"Thank you for doing this call I appreciate it. I was wondering if you could be specific about what you will not be able to do if Congress does not pass the American Rescue Plan. And separately, I'm interested in hearing from Dr. Walensky how accurate is the CDC data showing that states and localities are not using about half the vaccine distributed to them?"
102
Question
Negative

00:40:25-00:40:28 (4 sec)

"And if that data is the wrong picture, what is the accurate picture?"
103
Jeff Zients
Very Positive

00:40:28-00:40:58 (30 sec)

"Thank you, Cheryl. I'll take the first question and then I'll turn it over to Dr. Walensky. It's essential that Congress pass the act. We are committed to 100 shots in 100 days and that piece of it is not dependent upon Congress providing funds. But as we all know, that's just the start. We need Congress to provide money from additional vaccinations sites that provide for more vaccinators."
104
Jeff Zients
Positive

00:40:58-00:41:23 (25 sec)

"So in order to get all Americans vaccinated, we need Congress to provide funds for vaccination. We still do too little testing in this country. We need to ramp up testing significantly. So we need Congress to fund more testing in order to reopen schools and businesses and take care of people in congregant settings."
105
Jeff Zients
Negative

00:41:23-00:41:42 (19 sec)

"Furthermore, believe it or not, we still have shortages of PPE and other critical materials. We need emergency funds in order to make sure that we have those materials. So those are just three of the key areas that need to be funded by Congress in order for us to execute on the president's national plan."
106
Jeff Zients
Positive

00:41:42-00:41:47 (5 sec)
"Maybe you should repeat the question for Dr. Walensky and I'll hand it over to her."
107
Question
Positive
00:41:47-00:42:01 (15 sec)
"Yes. The question for Dr. Walensky is if -- how accurate is your data showing that states and localities are using only half the vaccine supply? And if that is not the real picture, then what is the accurate picture?"
108
Rochelle Walensky
Very Positive
00:42:01-00:42:27 (26 sec)
"Yeah, it's a great question. We have a database that actually looks at the vaccine supply. I want to reiterate that, you know, not all vaccine that is sort of allocated or delivered or whatnot is available for inserting into people's arms and that where in the pipeline that is varies by the day of the week, whether it's available that singular day."
109
Rochelle Walensky
Positive
00:42:27-00:42:53 (26 sec)
"We are working very hard in ensuring we understand exactly where the vaccine is on any given day and the -- the reporting of the data is actually also behind. So we're getting when vaccines are reported rather than when they are actually administered. So much of our work over the next week is going to make sure that we can tighten up the timelines to understand where in the pipeline the vaccine actually is and when exactly it is administered."
110
Jeff Zients
Very Positive

00:42:53-00:43:19 (26 sec)

"Yeah, let me add two points. One, most states are getting better at putting needles in arms and the federal government is beginning to support those efforts through all of the vehicles that Andy described in partnership with the states. Some of what is the states are -- have right now is inventory to do the very, very important second shot."
111
Jeff Zients
Very Positive

00:43:19-00:43:41 (22 sec)

"So I think it's important that when you're looking at states' inventories that you recognize that some of that inventory is Feeding health at the very important second shot, which we all believe that everyone should follow the FDA guidelines, as Dr. Walensky said earlier. So states are getting better. The federal government is ramping up its support."
112
Jeff Zients
Somewhat Positive

00:43:41-00:43:47 (6 sec)

"We've got a long way to go and we're very committed to the 100 million shots in the first 100 days."
113
Operator
Very Positive

00:43:47-00:43:53 (6 sec)
"Great. Let's go next to Ed O'Keefe at CBS News."
114
Question
Positive

00:43:53-00:44:17 (24 sec)
"Thank you all for doing this. A few kind of questions about Defense Production Act and the aspects of sort of how the administration can maybe hasten the production of vaccine related products. Regarding these new doses from Moderna and Pfizer you all announced yesterday -- those two companies specifically tell you when these extra 100 million doses will be available."
115
Question
Very Positive

00:44:17-00:44:39 (22 sec)
"I know some administration official said yesterday over the course of the summer, Moderna said third quarter of 2021. That's a broad range so I'm curious if you can narrow that down? And why it isn't being used yet the DPA, to perhaps compel other companies that make other pharmaceuticals to help make approved vaccines?"
116
Jeff Zients
Neutral

00:44:39-00:44:52 (13 sec)
"So let's each take -- taking the question one at a time. So Andy, why don't you start with the DPA and then also touch on the likely delivery of the additional 200 million doses?"
117
Andy Slavitt
Very Positive

00:44:52-00:45:16 (24 sec)

"Great. Thanks for the question. The Defense Production Act is a very important vehicle for us and we have an entire team that's focused on how to maximize and speed supply. Now, in its first instance, getting the syringes and all of the other ancillary capabilities necessary to get Americans their vaccines is one of the core areas."
118
Andy Slavitt
Very Positive

00:45:16-00:45:37 (21 sec)

"We've identified 12 areas where the president has authorized us to use the Defense Production Act including masks, including more testing, including many spaces. To the very specific question of whether or not a factory can be retrofitted to mass produce another vaccine -- that's something that's under active exploration."
119
Andy Slavitt
Slightly Negative

00:45:37-00:46:11 (34 sec)
"As you can imagine, it's not as simple as walking in and flipping a switch. These are delicate processes, very sterile processes and have to be done in a way that actually completely works. But we will not be afraid to explore every option to get more vaccines to the public as quickly as possible. As for the timing of Moderna and Pfizer contracts you know, I think look, it is no secret it is our goal to accelerate that as much as possible to the extent that we run into those physical limitations."
120
Andy Slavitt
Very Positive

00:46:11-00:46:23 (12 sec)
"We have to make sure that the -- that the vaccines that are produced are done the right way so that we can sign off and get them to the states and get them to the public. So right now, that looks like sometime over the summer."
121
Question
Somewhat Positive

00:46:23-00:46:36 (13 sec)
"So hypothetically then what you are saying you are exploring the possibility of saying again this is hypothetically potentially having someone like Merck produce the Moderna vaccine by using the DPA to do that?"
122
Andy Slavitt
Very Negative

00:46:36-00:47:05 (29 sec)
"Well I don't want to -- I don't want to talk about specific names per se, but I will say that you could -- we are exploring every possible option. You know, we do not believe that in a emergency crisis situation that we are in that it is responsible to just pick one path and pin our hopes on that. Obviously, there are other vaccine manufacturers that are submitting Emergency Use Authorization applications."
123
Andy Slavitt
Very Positive

00:47:05-00:47:25 (20 sec)
"There are other manufactures that are not pursuing their own vaccine. And for all of those we will be looking at ways to accelerate. When we make a commitment to the public, we don't want to make a commitment to the public based on things that have not been accomplished. We want to make a commitment based on things that we know about today."
124
Andy Slavitt
Neutral

00:47:25-00:47:31 (6 sec)

"And that is why we are talking about over the course of the summer we will do everything in our power, of course to do that."
125
Jeff Zients
Very Negative

00:47:31-00:47:56 (25 sec)

"To summarize it, this is a national emergency, 400,000 people have died Everything is on the table across the whole supply chain. Syringes, to other pharmaceutical companies, to anything we can do to increase the vaccine supply in the timing of the delivery is on the table. And we will execute accordingly."
126
Operator
Very Positive

00:47:56-00:48:02 (6 sec)

"Great. Let's go to Kristen Welker NBC News."
127
Question
Very Positive

00:48:02-00:48:30 (28 sec)
"Thank you so much. Couple of questions. Just to follow up on and I just want to be very clear -- has the president actually signed the DPA? And can you speak to the timeline you raised the issue of summer? And just so we're clear on the expectation by the end of summer. President Biden said that he was confident by the end of the summer 300 million people would be vaccinated."
128
Question
Very Positive

00:48:30-00:48:50 (20 sec)
"Is that still your expectation? And then can you speak to the concerns we have heard from governors who have cheered this announcement and said we welcome more supplies, we welcome more vaccines but in the words of Larry Hogan quote, we are going to need much more supply -- are you able to meet what the governors are asking for?"
129
Question
Neutral

00:48:50-00:48:55 (5 sec)
"Not just increasing it in this first chunk but continue to give what they are asking for?"
130
Andy Slavitt
Very Positive

00:48:55-00:49:15 (21 sec)

"Thank -- thank you for the question. Let me -- let me speak to that. First of all, I think it's important to clarify the difference between 300 million or 600 million vaccines being produced and the process of getting that across the country to providers and pharmacies, community clinics sand into people's arms."
131
Andy Slavitt
Very Positive

00:49:15-00:49:43 (27 sec)
"There are many more challenges. So what we've talked about yesterday, what the president announced was a first step, mainly to make sure we have enough production. Making sure that 300 million people get vaccinated have a lot -- have a lot of other elements. Including, community conversations to discuss people's concerns about vaccines as we get into the summer including making sure that to our earlier points we are getting every community access to vaccinations."
132
Andy Slavitt
Very Positive

00:49:43-00:50:09 (26 sec)
"So there is a huge process which we are over -- undertaking to do this. And so, I don't want you to hear that just because the -- they are -- will be manufactured and produced by a certain date that that is the same thing as taking every step necessary to get them to the public. As for Governor Hogan's comments about wanting more vaccines -- we hear you, Governor Hogan and we couldn't agree more."
133
Andy Slavitt
Very Positive

00:50:09-00:50:35 (27 sec)
"I would love to tell you that we inherited a situation where there were stockpiles and stockpiles of vaccines sitting there. That is not the case. And it is our job to level both with the governors and with the public. Having said that -- what we have been able to do is make a commitment to increase by 16 percent the amount of vaccines going out and to provide three weeks of visibility which will have led the plan better."
134
Andy Slavitt
Very Positive

00:50:35-00:50:41 (6 sec)

"And we hope to do our best to increase and accelerate more vaccines as we see them."
135
Operator
Very Positive

00:50:41-00:50:48 (7 sec)
"Great. Let's go to Yamiche Alcindor PBS."
136
Question
Very Positive

00:50:48-00:51:07 (19 sec)
"Hi. Thanks so much for taking my question. Two questions. First is, [Inaudible] sure how many vaccinations are currently available and what's left in the federal stockpile? Is there -- are we dealing with a finite number of vaccinations or are they coming in a certain -- I'm just wondering if you could talk through the numbers again."
137
Question
Very Positive

00:51:07-00:51:22 (15 sec)
"And second question I have is -- what percentage of vaccinations, that are already vaccinated is coming back with racial ethnicity information? Because I'm wondering how we know who is getting what, what you're gaging success when it comes to the people who are getting vaccinated?"
138
Jeff Zients
Somewhat Positive

00:51:22-00:51:30 (8 sec)

"So Andy, why don't you take the first question. Dr. Nunez-Smith the second, please."
139
Andy Slavitt
Very Positive

00:51:30-00:51:55 (26 sec)
"Yes. So today we've delivered 47 million vaccines to states. And we are committed to delivering an additional 10 million per week for the next three weeks to states. Now, we do not keep a stockpile of a large number of vaccines. We keep a two to three day supply as a practice just to account for variation in manufacturing production."
140
Andy Slavitt
Neutral

00:51:55-00:51:58 (2 sec)
"That's it. The rest moves out to states."
141
Question
Positive

00:51:58-00:52:04 (6 sec)

"So thanks for [Inaudible]"
142
Jeff Zients
Slightly Negative

00:52:04-00:52:29 (25 sec)
"I think part of the confusion here is there was a practice early on before we were in office of having a stockpile. The [Inaudible] days as Andy said of the supply to deal with any unforeseen situations. Dr. Nunez-Smith."
143
Marcella Nunez-Smith
Very Positive

00:52:29-00:52:59 (30 sec)

"Great. Thank you. And thank you for that -- that question. So we are 100 percent committed to making sur that when it's your turn that you have access sot the vaccine. And we know that access in -- in many communities that have been hard hit might be a challenge. You know, to your specific question around race, ethnicity data -- you know, we are making a call to get better, more consistent data."
144
Marcella Nunez-Smith
Slightly Positive
00:52:59-00:53:21 (22 sec)

"Particularly around some of these demographic points. You know, we see right now around 16 states or so that are releasing race ethnicity data. But it's true that those data are incomplete. But it's not our only you know, sort of visibility into what's happening. The -- the CDC for example, has a metric around social vulnerability that you know, many states are using that."
145
Marcella Nunez-Smith
Very Positive

00:53:21-00:53:41 (20 sec)

"Now we encourage the use of equity metrics such as that as states are doing their mapping and planning. So we can think about things like -- like neighborhoods and communities as metrics and ways to track as well. And we're -- we're building our -- our equity dashboard. Right now, we will rely on government sources as well as -- as sources of data external to government."
146
Question
Somewhat Negative

00:53:41-00:53:59 (19 sec)
"And just a follow up -- how soon do you think you will plan to roll out the vaccine to communities of color have a real impact? I know we talked about summer and -- and other months for the nation as a whole. I wonder if there are any estimates for communities of color and more vulnerable populations?"
147
Marcella Nunez-Smith
Positive
00:53:59-00:54:22 (23 sec)

"Absolutely. So you know that work is happening right now. So in states and localities you know, where you know, depending on -- on kind of what's on the ground as are as prioritization. But in those communities that have been hard hit you know, I say kudos to all of the local health officials who are making those pushes to get to those who are over 75 you know, who are in affordable housing communities."
148
Marcella Nunez-Smith
Neutral
00:54:22-00:54:41 (19 sec)

"Who are bringing vaccine to people you know, in homeless shelters. You know who, again, meet the criteria for the prioritization. So I think that's the key is when we say you know, everyone over a specific age is eligible. We are going to have to take an extra step to get to people who are hardest to reach."
149
Marcella Nunez-Smith
Neutral

00:54:41-00:54:42 (1 sec)

"And that work is already happening there."
150
Question
Somewhat Positive
00:54:42-00:54:45 (3 sec)

"Thank you."
151
Jeff Zients
Slightly Positive

00:54:45-00:54:51 (6 sec)

"And we're coming up on the top of the hour so we have time for more. We go to Alice Park at Time."
152
Question
Very Positive
00:54:51-00:55:15 (24 sec)
"Hello and thank you so much for taking our question. So this is more for the scientist Dr. Fauci and Dr. Walensky. It seems that the mutation on SARS-COVID-2 are inevitable and it's really a matter of staying on top of it and figuring out which ones are gonna be benign and which ones potentially affect disease outcome and ultimately vaccine immunity."
153
Question
Neutral
00:55:15-00:55:33 (18 sec)
"Are there plans on the current sequence of efforts on a national level that provides states with additional funding and collecting samples and sequencing themselves? And how high of a priority is this to have a more national sequencing effort for surveillance purposes?"
154
Jeff Zients
Somewhat Positive
00:55:33-00:55:36 (4 sec)
"Dr. Walensky, would you like to take that first?"
155
Rochelle Walensky
Very Positive
00:55:36-00:56:06 (29 sec)
"Thank you. Yes, we are actively working on this. These are data and samples that are collected in the states as well as with academic and pharma collaborations. I will highlight again, there is money in the American recovery act to move this forward. And we really need to be -- have access to those resources to do the amount of sequencing and surveillance that we need in order to be able to detect these when they first start to emerge."
156
Jeff Zients
Very Positive

00:56:06-00:56:48 (43 sec)
"Just -- just to add and expand -- believe it or not, we are 43rd in the world at genomic sequencing. So it's essential as part of the American Rescue Plan [Break in Audio] variants I'm getting signal I'm going in and out. Can you hear me now? All right, so I'll back up for a second we are 43rd in the world for genomic sequencing."
157
Jeff Zients
Very Positive

00:56:48-00:57:04 (16 sec)

"Totally unacceptable. As part of the American Rescue Plan we remedy that situation and [Inaudible] appropriate amount of genomic sequencing which will allow us to spot variants early which is the best way to deal with any potential variants. Dr. Fauci?"
158
Anthony Fauci
Very Positive
00:57:04-00:57:27 (23 sec)
"One other thing I -- I think people need to understand is that as we all know, RNA viruses mutate all the time. That's -- that's what they do. That's their business. And there are very few but they do happen occurring that is that you get one that has a functional relevancy to it. The way we've seen with greater transmissibility, possibility of greater virulents."
159
Anthony Fauci
Somewhat Positive
00:57:27-00:58:00 (33 sec)
"But also, importantly for us, what I mentioned in my opening comments is that what is the relationship between the mutant and the induction of antibodies by the vaccines that we do. So supplementing what Dr. Walensky said, that together with the CDC's expansion in getting more real time sequence genomic surveillance the NIH will be collaborating with the CDC in looking at what the functional characteristics of these are."
160
Anthony Fauci
Neutral
00:58:00-00:58:27 (27 sec)
"For example, we will be monitoring in real time the effect of antibodies that we induce with the current vaccines and with future vaccines as to what impact they have on the ability to neutralize these mutants. And as we see them getting further and further to a more vulnerable part that's when we trigger the kinds of things that I mentioned in my opening remark."
161
Anthony Fauci
Neutral
00:58:27-00:58:43 (16 sec)
"Namely, making a version of the same vaccine that in fact, would be directed specifically against the relevant mutant. And all of that is going on in real time literally as we speak."
162
Jeff Zients
Very Positive

00:58:43-00:59:25 (41 sec)

"Okay. I think we're going to wrap it up. I want to thank everybody for joining. As you can see lots of information on the disease front, the CDC, science, Dr. Fauci and the operations from Andy Slavitt [Inaudible]. We plan on doing these on a regular basis, and the next briefing will be on Friday. Thank you for your patience and for all your great questions."
163
Jeff Zients
Positive

00:59:25-00:59:28 (4 sec)
"Appreciate it. "
164