Fact
ba
.
se

Press Conference: White House COVID-19 Response Team Holds a Briefing - January 29, 2021
-
Anthony Fauci Person
-
Andy Slavitt Person
-
Rochelle Walensky Person
-
Question Person
-
Unidentified Person
Andy Slavitt
Very Positive

00:00:00-00:00:30 (30 sec)
"Good morning. Thank you for joining us. I am Andy Slavitt, senior advisor with the White House. There's been a lot happening over the last week and over the last 24 hours, and we appreciate you joining these regular briefings. We are working hard on executing our plan, which begins with a frank and open dialogue with the American public, so there is clarity into what we see, what we are working on, what actions we are taking, and on both the successes and the hurdles we face."
1
Andy Slavitt
Somewhat Positive

00:00:30-00:00:57 (28 sec)
"I hope on these calls that the public will get a regular and consistent dose of what the CDC believes are the right public health actions on an array of challenging topics and how our leading infectious disease expert Dr.Fauci is advising the president and the country. I will have a couple of remarks, but first, I will turn it over to director of the CDC, Dr. Walensky."
2
Rochelle Walensky
Very Positive

00:00:57-00:01:16 (19 sec)
"Thank you. I am glad to be back with you all today. Good morning. To give you the latest update on the state of the pandemic. Let's begin with an overview of the data. Despite some encouraging trends in COVID-19 cases and hospital admissions, the occurrence of COVID-19 remains extraordinarily high in the United States."
3
Rochelle Walensky
Positive

00:01:16-00:02:31 (75 sec)
"We continue to have over four times the daily number of cases as we had over the summer. Through January 27, 25.4 million COVID-19 cases have been reported to CDC. During the week of January 21 to January 27, the seven-day average of new cases decreased 16.6 percent to 161,832 per day. The seven-day average of new hospital admissions of patients with COVID-19 during the week of January 20 to January 26 decreased by 14 percent to 12,720 per day; however, over 99,000 people were hospitalized with COVID-19 on January 26. 427,626 deaths have been reported since January 22 -- through January 22, 2020. During the week of January 21 to January 27 the seven-day average number of deaths increased 7.5 percent to 3,277 per day, and yesterday we saw our sixth day of over 4,000 deaths."
4
Rochelle Walensky
Very Positive

00:02:31-00:03:16 (45 sec)
"We continue to remain concerned about the emergence of variants in the United States and are rapidly ramping up surveillance and sequencing activities as we work to closely monitor and identify variants as they emerge. Any information we have, we will then share with you as rapidly as possible. The B117 variance that was first detected in the UK has now been confirmed in 379 cases in 29 states as of January 27. Yesterday South Carolina public health officials notified the public about the first two documented cases of the B1351 variant, which was officially -- first detected in South Africa, now in the United States."
5
Rochelle Walensky
Neutral

00:03:16-00:03:40 (24 sec)
"These cases were identified -- identified in different parts of the state and not believed to be epidemiologically linked. They each did not have any travel history. We are still learning more about their exposures, and earlier this week, Minnesota identified the first U.S. case of the P1 variant. This is the variant that originally emerged in Brazil."
6
Rochelle Walensky
Very Positive

00:03:40-00:04:03 (23 sec)

"CDC will continue communicating with international, state, and local partners to monitor the presence and impact of variants in the United States and around the world, and we are actively working with national reference laboratories, state health departments, and researchers across the country to improve our understanding of these variants and how it may impact the pandemic."
7
Rochelle Walensky
Very Positive

00:04:03-00:04:24 (21 sec)

"We also know viruses mutate, and they tend to mutate in ways that are advantageous to the virus. We expected this, and this is why I feel compelled to underscore for you the need for each of us to remain steadfast in our commitment to taking all of the appropriate steps to protect ourselves and our communities."
8
Rochelle Walensky
Somewhat Positive

00:04:24-00:04:48 (24 sec)

"You are going to hear me say this a lot, so here it is, wear a mask, stay 6 feet apart, avoid crowds and poorly ventilated spaces. Also, now is not the time to travel. If you choose to travel, please follow the CDC guidelines and be aware that you must wear a mask as you travel, and when it is your turn, please roll up your sleeve and get vaccinated."
9
Rochelle Walensky
Very Positive

00:04:48-00:05:19 (31 sec)
"If we do all of these things, there is less virus spreading, and the conditions can -- that produce virus -- variants are lessened, and before I turn it over to Dr. Fauci, I would like to just address school reopening's which I know has also been in the news a lot this week. CDC continues to recommend that K-12 schools be the last setting to close after all other mitigation measures have been employed and the first to reopen when they can do so safely."
10
Rochelle Walensky
Neutral

00:05:19-00:05:49 (30 sec)
"Accumulating data suggest school settings do not result in rapid spread of COVID-19 when the mitigation measures are followed, including masking, decreasing density, and proper ventilation. Implementing community-based strategies that reduce transmission when there is a lot of virus in a particular area is also important to supporting the safe reopening of schools, but we recognize many communities lack sufficient capacity to do all that is needed."
11
Rochelle Walensky
Very Positive

00:05:49-00:06:08 (19 sec)
"That is why it is essential that the guidance, tools, and resources called for in the national strategy for the COVID-19 response and the American rescue plan are provided to communities. Thank you. I look forward to taking your questions in a few minutes, and I will turn it over to Dr. Fauci. Dr. Fauci?"
12
Anthony Fauci
Very Positive
00:06:08-00:06:42 (34 sec)
"Thank you very much, Dr. Walensky. Well, as alluded to by Andy Slavitt, this past week and particularly the past 24 hours have been really interesting, and I think value-added to the issue of vaccine protection in the government attempt to contain this outbreak. In previous briefings, I have mentioned that the federal government had been involved in the development and/or facilitation of three separate platforms of vaccines."
13
Anthony Fauci
Positive
00:06:42-00:07:10 (28 sec)
"One was the mRNA, the other were vector, either an adeno or a chimp vector, and the other was the soluble recombinant proteins. We all know now of the recent data that came with regard to Moderna and Pfizer, with their mRNA showing a 94 percent to 95 percent efficacy and a very, very good efficacy against advanced or severe disease."
14
Anthony Fauci
Very Positive
00:07:10-00:07:49 (38 sec)
"Over the last 24 hours, there have been the announcements of two other trials that represent two additional platforms. One is the soluble protein platform in a study that came out of the UK using from the company Novavax and the other one that we discussed this morning at a press conference at the NIH was the Janssen or Johnson & Johnson adeno 26 trial that took place in the United States, in South Africa and in Brazil and the results really are very encouraging."
15
Anthony Fauci
Very Positive
00:07:49-00:08:18 (30 sec)
"Let me just very briefly outline for them -- for you and then maybe make a couple of comments regarding nuances of the study. In the study that was just reported by Janssen, Johnson & Johnson, it was a trial using their Ad26, and the overall vaccine efficacy in the study was 66 percent, but for the United States, it was 72 percent."
16
Anthony Fauci
Somewhat Negative
00:08:18-00:09:05 (47 sec)
"Now the important issue because the first thing people do is compare a 72 percent efficacy with the previously reported in other trials of 94 percent to 95 percent. That is true, but when one looks at the potential impact on a very important aspect of what we look at carefully is namely severe disease that overall in the United States, in South Africa, and in Brazil, the overall efficacy for severe disease was 85 percent, and in fact, in the study including in the South African isolate there were essentially no hospitalizations or deaths in the vaccine group whereas in the placebo group there were."
17
Anthony Fauci
Neutral
00:09:05-00:09:42 (37 sec)
"So this really tells us that we have now a value-added additional vaccine candidate that will, of course, as happens with every candidate will present the details of their data to the FDA. But I want to point out that this has important potential and real implications both domestically and globally because, as many of you are aware of this is a single-shot vaccine in which you start to see efficacy anywhere from seven to ten days following the first and only shot."
18
Anthony Fauci
Very Positive
00:09:42-00:10:19 (37 sec)

"It is very, very good with regard to cold chain requirements, namely requiring only a refrigerator. It is inexpensive, and the company is capable of making doses in the numbers of billions. Of note, and this is something that relates to what Dr. Walensky just said, we are being faced with variants. We are all aware of the variants that we knew dominated in the UK, the 117, the 351 in South Africa, and other variants such as the P1 in Brazil."
19
Anthony Fauci
Negative
00:10:19-00:11:00 (40 sec)

"When these variants were first recognized, it became clear that we had to look at in vitro in the test tube whether the antibodies that were induced by the vaccines that we had available would actually neutralize these new mutants, and it was reported in several pre-print journals over the past couple of weeks that although it diminished particularly the troublesome South African isolate, although it diminished the efficacy of the antibodies it still was not below the cutoff where you would expect some degree of efficacy."
20
Anthony Fauci
Leans Negative
00:11:00-00:11:26 (26 sec)
"But what we know now from this study, namely the J&J and the Novavax study that antigenic variation i.e. mutations that lead to different lineage, do have clinical consequences because, as you can see, even though the long-range effect in the sense of severe disease is still handled reasonably well by the vaccines."
21
Anthony Fauci
Very Negative
00:11:26-00:12:04 (38 sec)
"This is a wake-up call to all of us that we will be dealing as the virus uses its devices to evade pressure particularly immunological pressure that we will continue to see the evolution of mutants. So that means that we as a government, the companies, all of us that are in this together will have to be nimble to be able to just adjust readily to make versions of the vaccine that actually are specifically directed towards whatever mutation is actually prevalent at any given time."
22
Anthony Fauci
Slightly Positive
00:12:04-00:12:47 (43 sec)
"And finally, this all tells us that it is an incentive to do what we have been saying all along, to vaccinate as many people as we can as quickly as we possibly can because mutations occur because the virus has a playing field as it were to mutate. If you stop that and stop the replication, viruses cannot mutate if they don't replicate, and that is the reason to continue to do what we are doing, namely, intensify our ability and our implementation to vaccinate as many people as possible as quickly as possible."
23
Anthony Fauci
Very Positive
00:12:47-00:12:51 (5 sec)
"So now I will hand it back to Andy Slavitt. Andy?"
24
Andy Slavitt
Very Positive

00:12:51-00:13:16 (25 sec)

"Thank you, Dr. Fauci. Before I turn it over to all of you for some questions, I would offer a few updates and a sense of the action and activities and our progress against executing our plan to defeat -- defeat COVID-19. One of the core elements of our plan is accelerating the process of vaccinating the country to protect people from COVID-19 as quickly and as safely as possible."
25
Andy Slavitt
Neutral

00:13:16-00:13:43 (27 sec)
"For the past week, our seven-day average number of shots administered was 1.2 million per day. We view this number of 1 million doses per day as a base to build from in the coming days, weeks, and months. In total, we have now delivered 48 million doses, and 26 million of those doses have been administered."
26
Andy Slavitt
Very Positive

00:13:43-00:14:23 (40 sec)
"As I have said before, we are facing two challenges. The first is increasing the supply of vaccines safely and more rapidly and speeding up the time it takes to administer them efficiently and importantly equitably. I want to call out seven states that have already provided first vaccinations to more than 10 percent of their adult populations: Alaska, West Virginia, New Mexico, Connecticut, North Dakota, Oklahoma, and South Dakota."
27
Andy Slavitt
Very Positive

00:14:23-00:15:02 (39 sec)
"Well done. So we have taken steps to announce last week that we will increase supply week over week by 16 percent and vastly improve the predictability of ordering vaccines by giving everyone a three week forward window into how many vaccines they will be getting delivered. And we announced plans to purchase 200 million additional vaccines, which means that no matter what happens with other approvals, we will have sufficient supply to vaccinate the country."
28
Andy Slavitt
Very Positive

00:15:02-00:15:40 (38 sec)

"We are also taking steps to speed the vaccination administration process. At the president's direction, FEMA has -- has increased ITS support to states, tribes, and territories for vaccination sites. FEMA is providing nearly $1 billion to support vaccinations sites in states and territories. More than 200 FEMA staff members are on the ground today providing logistical support in eight states and the agency is providing federal equipment and supplies to support states across the country."
29
Andy Slavitt
Neutral

00:15:40-00:16:06 (26 sec)
"In addition, we are getting more vaccinators into the field to continue to escalate the pace of vaccinations. Yesterday, the Department of Health and Human Services amended the PREP Act declaration to permit recently retired doctors and nurses to administer COVID-19 vaccines and to permit anyone currently licensed to vaccinate within their home state."
30
Andy Slavitt
Very Positive

00:16:06-00:16:31 (25 sec)
"That's doctors, nurses, EMTs to be able to administer shots across state lines. We are looking at every possible step to make it easier to get more vaccinations out in the field. We encourage individuals to contact their local and state health departments, their local medical reserve core unit to sign up to volunteer if they are qualified."
31
Andy Slavitt
Very Positive

00:16:31-00:16:52 (21 sec)
"And today, the acting Health and Human Services secretary will continue the activation of the U.S. Public Health Service Commission Corps. He's requesting public health service officers from all available categories to support national COVID-19 vaccination efforts. I want to also touch on our efforts to make the workplace safer."
32
Andy Slavitt
Very Positive

00:16:52-00:17:21 (29 sec)
"The Department of Labor announced that its Occupational Safety and Health Administration, or OSHA, has issued stronger worker safety guidance to help employers and workers implement a coronavirus mitigation program and better identify risks which could lead to exposure and infection. Ensuring the health and safety of all of our country's workers is a national priority and a moral and economic imperative."
33
Andy Slavitt
Somewhat Positive

00:17:21-00:17:50 (29 sec)
"Healthcare workers and other essential workers, many of whom are people of color, immigrants have put their lives on the line during the coronavirus pandemic and this updated guidance provides a road map for businesses to protect the health of their workers. Now, one of the purposes of these briefings is to demonstrate the transparency that's within our national plan."
34
Andy Slavitt
Very Positive

00:17:50-00:18:20 (29 sec)
"And so I want to close by mentioning some of the steps we are taking to improve public access to the information they should have at their fingertips to stay safe. This week, we released previously nonpublic data on the pandemic trends across all 50 states in detail. This detailed information was previously reserved solely for governors and it is now available to everybody to see."
35
Andy Slavitt
Very Positive

00:18:20-00:18:45 (25 sec)

"These will be weekly reports publicly posted online. So our team is working hard to make the process more transparent and easier for the public to understand beginning with these briefings and the very clear messages from Drs. Walensky and Fauci. So let me turn and spend the balance of our time taking questions for Dr. Walensky, Dr. Fauci, and myself."
36
Unidentified
Very Positive

00:18:45-00:19:00 (15 sec)

"Great. Thanks, Andy. And right now, we have time for a couple of questions. If you have not already, you can go ahead and raise your hand on the Zoom feature. First up, we're going to go to Christopher Roland at the Washington Post."
37
Question
Very Positive

00:19:00-00:19:34 (34 sec)

"Yes, thank you. Thanks for doing the briefing. On the subject of the low dead space syringes, I was wondering what percentage of the administration kits now are being sent out with the Pfizer vaccine contain those specialized units, how many more millions more do you need to get to 100 percent, and what companies are being ordered to procure more under the DPA? And also on the state allotments on and Pfizer's credit, are state allotments based on five doses in the vial or six?"
38
Question
Somewhat Positive

00:19:34-00:19:37 (3 sec)

"And is Pfizer getting credit for all six doses?"
39
Andy Slavitt
Very Positive

00:19:37-00:19:59 (22 sec)
"Thanks for the question. And at least at risk of somebody coming back and correcting my answer, I believe the answer is 100 percent and they're getting credit for six precisely because everybody should now have the tools to be able to get the sixth Pfizer dose out. That's it thanks to the work of our DPA and supply chain team."
40
Andy Slavitt
Neutral

00:19:59-00:20:01 (2 sec)

"Next question."
41
Unidentified
Very Positive

00:20:01-00:20:07 (6 sec)

"Great. Next, it will go to Kaitlan Collins at CNN."
42
Question
Very Positive

00:20:07-00:20:30 (23 sec)
"Thank you. I have two questions, actually. One on these new variants that we're seeing and the expectation seems to be that they could really start to have an impact on cases by March, maybe gain a foothold by April and become the dominant cases that we are seeing. So what specifically is the administration doing to stop that from happening beyond recommending that people social distance and wear masks?"
43
Andy Slavitt
Very Positive

00:20:30-00:20:50 (20 sec)

"Well, let me turn first to Dr. Fauci to talk about the science behind these variants and what -- what is likely to happen or what we know and what we believe. And then to Dr. Walensky after that so she can discuss the actions we are taking as a country and the actions we expect Americans to take to help -- help with your -- your question."
44
Andy Slavitt
Somewhat Positive

00:20:50-00:20:53 (2 sec)

"Thank you."
45
Anthony Fauci
Very Positive
00:20:53-00:21:36 (43 sec)
"Well Caitlin, there are a number of variants that we are concerned about. One that is quite well established already in the United States and that is the 117 that is in about 28 or 29 states and more than 315 cases that have all been reported. As you alluded to, the fact is that when you have a virus that has ability to transmit more efficiently than the wild type in the communities, sooner or later by pure viral dynamics itself, it will become more dominant than the wild type."
46
Anthony Fauci
Positive
00:21:36-00:22:05 (30 sec)
"So we have that already there. We have a situation where they have now been reported in very specific places in South Carolina, for example, the isolate or that mutant that is the 351 from -- dominant in South Africa. Again, that seems to have a very good fitness for spread. So whether or not that's going to ultimately take over in the sense of being dominant is unclear by now."
47
Anthony Fauci
Very Positive
00:22:05-00:22:48 (43 sec)
"The projection that is made with regard to that UK is that probably by the end of March, at the beginning of April it actually will become more dominant in this country. As I mentioned at the very end of my remarks, I'll leave it to -- in a moment to Dr. Walensky to talk a bit about what we're doing from a surveillance another standpoint, but the fundamental principle of getting people vaccinated as quickly and as efficiently as you possibly can will always be the best way to prevent the further evolution of any mutant because when you do that, you prevent replication and replication is essential for mutation."
48
Anthony Fauci
Neutral

00:22:48-00:22:50 (2 sec)
"So over to you, Dr. Walensky."
49
Rochelle Walensky
Leans Positive
00:22:50-00:23:14 (23 sec)

"Thank you, Dr. Fauci, and for that question. So there are numerous things that we are doing. As Dr. Fauci noted, the -- the prevention measures and the mitigation measures for variants are actually exactly the same, regardless of what variants you have. We have scaled up surveillance dramatically just in the last 10 days, in fact, but our plans for scaling up surveillance are even more than what we've done so far."
50
Rochelle Walensky
Leans Positive

00:23:14-00:23:41 (28 sec)
"So we have partnerships with commercial lands. We are now asking surveillance from every single state, at least 750 surveillance strains per week and we have 7 collaborations across universities to scale up surveillance to the thousands per week so that we can get a foothold to see whether these projections of having this take hold rather than by the middle of March or late -- middle to late March are actually true."
51
Rochelle Walensky
Very Positive

00:23:41-00:24:04 (22 sec)
"What we're also going to be doing is offering support to the states that have identified these surveillances, specifically South Carolina with the 351 variant reaching out to help with vaccination as vaccination is, again, one of these things we need to do with resources for increased testing, increased surveillance, as well as increased resources for vaccination and funding."
52
Rochelle Walensky
Slightly Positive

00:24:04-00:24:27 (23 sec)

"So we are working hard to make sure that we can actually do the mitigation measures, the vaccination, do the surveillance. And then finally, we have cross agency collaborations with CDC, NIH, BARDA, DoD so that when we get these strains, we can do the science and understand the impact of these variants on our vaccines as well as on our therapeutics."
53
Andy Slavitt
Very Positive

00:24:27-00:24:58 (31 sec)
"Let me add one more comment. Look, if we as a country want to turbocharge our efforts at sequencing, which I believe should be shared bipartisan perspective, we can do that. And what we need is the Congress to quickly pass the American Rescue Plan, which contains the resources necessary to get all of our very, very talented people around the country who are world experts in sequencing to get on this as quickly as possible."
54
Andy Slavitt
Very Positive

00:24:58-00:25:09 (11 sec)

"So I couldn't urge people to -- people in the Congress to be more -- more focused on the American rescue plan than I can, given the state of the variants."
55
Rochelle Walensky
Very Positive

00:25:09-00:25:33 (25 sec)

"Maybe if I could make one more point, and that is that by the time someone has symptoms, gets a test, has a positive result, and we get the sequence, our opportunity for doing real case control and -- and contact tracing is -- is largely gone. And so I think and I believe that we should be treating every case as if it's a variant during this pandemic right now."
56
Andy Slavitt
Very Positive

00:25:33-00:25:36 (3 sec)
"Thank you. Thank you for the question. Next."
57
Unidentified
Very Positive

00:25:36-00:26:13 (37 sec)

"Great. Now we'll go to Ed O'Keefe at CBS. Ed, I think you can speak. All right, Ed, we'll -- we'll try to come back to you. In the meantime, let's go to Caroline Chen."
58
Question
Very Positive

00:26:13-00:26:45 (32 sec)
"Hi. Thanks for taking my question. This is a question for both Dr. Walensky and Dr. Fauci. So when epidemiologists talk about herd immunity, they often say about 70 or 80 percent of the population is the goal. Given that about 20 percent of the U.S. population is children, do you think it's important to have eligibility expanded to children for vaccination as soon as possible and, if so, what are -- what is being done to -- to get to that goal?"
59
Anthony Fauci
Very Positive
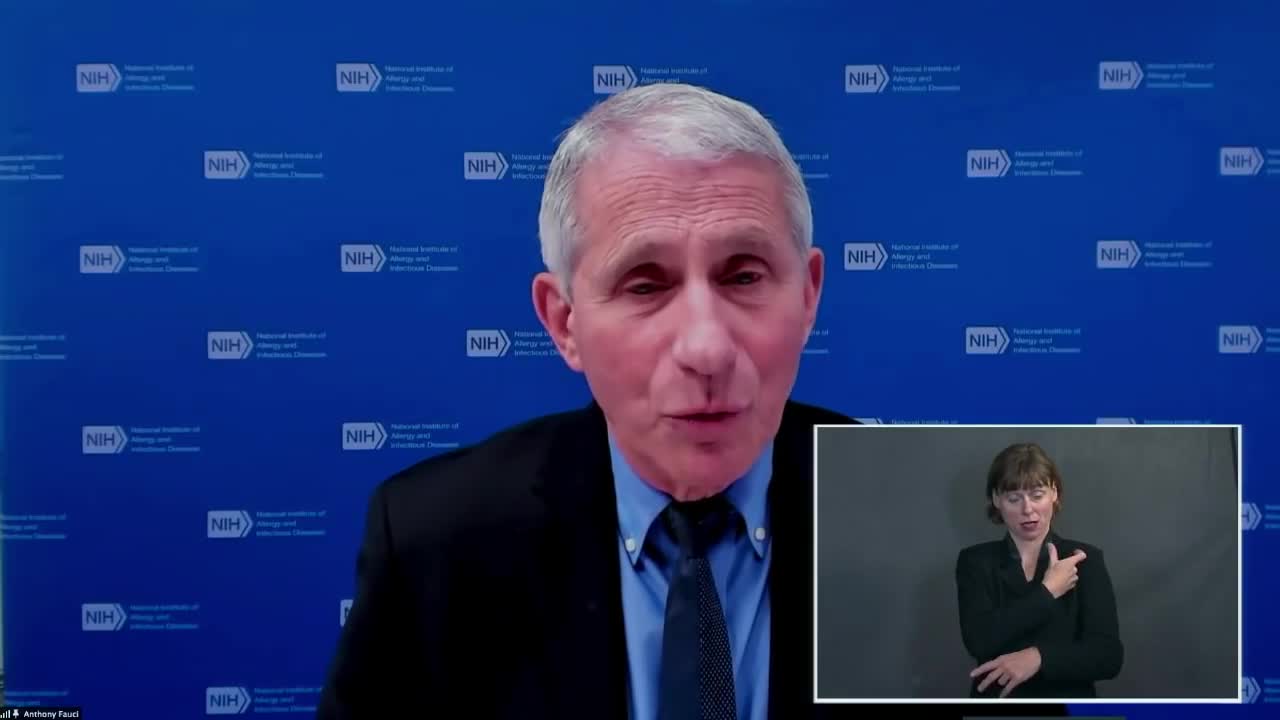
00:26:45-00:27:09 (25 sec)
"Yes, we're -- that's a very good question and in the candidates that we're talking about now, certainly the Moderna and the Pfizer, the two that have been given the EUA, we've already started on what's called an aged de-escalation testing. But that means that you start off, for example, we know with one of the vaccines at 16 and then one is 18 years old."
60
Anthony Fauci
Positive
00:27:09-00:27:38 (29 sec)
"We go down from 16 to 12, from 12 to 9, and what you do, because you don't want to have to, and I'll explain why in a moment, to go through an efficacy trial where you're involving tens of thousands of children to show efficacy, what you can do is in a much smaller trial, measure it in hundreds to a couple of thousands to do what we call safety in a phase 2A for immunogenicity."
61
Anthony Fauci
Positive
00:27:38-00:28:01 (23 sec)
"And if you can show that it's safe and that, in fact, it induces the kind of response that is reflective of the protection, namely the correlant of immunity, what you can do is then bridge that to the efficacy data that you got from the 30,000 trial with Moderna and the 44,000 trial that we did with Pfizer."
62
Anthony Fauci
Neutral
00:28:01-00:28:21 (20 sec)

"So in direct answer to your question, over the next couple of months, we will be doing trials in a -- in an aged de-escalation manner so that, hopefully, by the time we get to the late spring and early summer, we will have children being able to be vaccinated according to the FDA's guidance."
63
Question
Somewhat Positive
00:28:21-00:28:22 (1 sec)

"Thank you."
64
Andy Slavitt
Neutral
00:28:22-00:28:25 (3 sec)

"Dr. Walensky, do you want to add anything?"
65
Rochelle Walensky
Neutral

00:28:25-00:28:47 (21 sec)
"I was just going to add our current data from schools, from summer camps and whatnot also suggested that the children not only have decreased rates of symptoms, but have decreased rates of transmissibility. Those estimates for herd immunity are very much based on rates of transmissibility, and so what pertains to herd immunity among adults may be different among children."
66
Andy Slavitt
Somewhat Positive

00:28:47-00:28:49 (3 sec)

"All right. Thank you. Next question."
67
Unidentified
Neutral

00:28:49-00:29:00 (10 sec)

"All right. We have time for two more. We are going to go to Sharon LaFraniere at New York Times."
68
Question
Somewhat Positive

00:29:00-00:29:01 (1 sec)

"Thank you. Can you hear me?"
69
Andy Slavitt
Positive

00:29:01-00:29:01 ( sec)

"Yes."
70
Question
Slightly Negative

00:29:01-00:29:22 (21 sec)
"So Dr. -- Dr. Fauci said that we're going to have to make new versions of the existing vaccine to combat the variants and the vaccine makers have shown that they can design vaccines, but where is the extra manufacturing capacity going to come from? Aren't the vaccine makers already tapped out making the vaccine?"
71
Question
Slightly Positive

00:29:22-00:29:27 (5 sec)

"So, what's the plan for making revised vaccines and booster shots on top of that?"
72
Andy Slavitt
Very Positive

00:29:27-00:29:49 (22 sec)

"Thanks for the question. I -- I think this question speaks to a larger issue because I think you could have asked that question with a number of different permutations, and that's the issue of contingency planning and making sure we have sufficient vaccines -- sufficient manufacturing capacity and enough space in our contracts to be able to make adjustments on the fly."
73
Andy Slavitt
Leans Negative

00:29:49-00:30:13 (23 sec)

"And I would tell you that the Department of Health and Human Services, the FDA, the -- the team led by Dr. Kessler are currently hard at work answering all of those questions. So, I will not use these briefings to talk about nonpublic information about specific companies. All I can tell you is those are exactly the right factors that we are thinking about."
74
Andy Slavitt
Very Positive

00:30:13-00:30:40 (27 sec)

"And I think the -- the -- the -- to make a broader point, the teamwork between everybody here, between the NIH, the CDC, the FDA, and then -- and then our ability to -- to understand what those factors are and turn it into actual actionable plans with the manufacturers is really important, and I have been incredibly impressed at least is so far with that."
75
Unidentified
Very Positive

00:30:40-00:30:46 (6 sec)
"Great. And we'll -- we'll send our last question to Yamiche at PBS."
76
Andy Slavitt
Slightly Negative

00:30:46-00:30:50 (4 sec)

"You cut off."
77
Question
Neutral

00:30:50-00:30:53 (3 sec)

"Hi. It's Yamiche Alcindor with PBS."
78
Andy Slavitt
Neutral

00:30:53-00:30:53 ( sec)

"Hi."
79
Question
Positive

00:30:53-00:30:57 (3 sec)

"Thanks so much for taking my question. Can you hear me?"
80
Andy Slavitt
Positive

00:30:57-00:30:58 (1 sec)
"Yes, I can."
81
Question
Very Positive

00:30:58-00:31:14 (17 sec)
"Okay. The -- I have two questions. The first is it sounded like you were saying that we're vaccinating about a million people daily. I wonder if you can talk about what the goal would be, the -- the average goal would be, and -- and how quickly we ramp up to that average. Like, what would be a better average?"
82
Question
Neutral

00:31:14-00:31:16 (2 sec)
"And then I have a second question after that."
83
Andy Slavitt
Very Positive

00:31:16-00:31:45 (29 sec)

"Well, so it -- our seven-day average is 1.2 million per day. As I said, we view where we are today as a floor, not a ceiling. What we have tasked our team with is as many vaccines as possible into as many arms as possible. There are two constraints right now. The first is just continuing week after week to press to increase productions and find opportunities as they exist."
84
Andy Slavitt
Very Positive

00:31:45-00:32:11 (26 sec)

"That is a slow process. That doesn't happen overnight. The second where I think there is opportunity is to turn those vaccines produced into actual vaccinations more rapidly, and we've announced a number of steps to do that. So, I'm not going to put out a different number today on what I'd like to see, other than to tell you that, every day when the number cames out -- comes out, all of us are breathlessly awaiting the number and looking for as high a number as possible, of course."
85
Andy Slavitt
Neutral

00:32:11-00:32:14 (3 sec)
"I -- I know you have another question, Yamiche."
86
Question
Neutral

00:32:14-00:32:34 (20 sec)
"And then I wanted to ask about the -- two-thirds of cases in Los Angeles County, it's -- it's been reported, came out in the last two months, or have been reported in the last two months. One fact -- one study said that the factor was this recent coronavirus surge of a new variant, CAL.20C. What more do we know about the LA variant?"
87
Question
Neutral

00:32:34-00:32:47 (12 sec)

"And what does that tell us about other big cities? Should -- should we expect maybe a New York, a -- or a New Orleans variant? How -- how -- how does -- what is LA teaching us about what our big cities might be experiencing?"
88
Andy Slavitt
Positive

00:32:47-00:32:49 (2 sec)

"Thank you. Dr. Fauci, do you want to take that one?"
89
Anthony Fauci
Very Positive
00:32:49-00:33:16 (27 sec)
"Yeah, sure. Well, what it tells us is what I alluded to in one of my prior comments, that when you have a significant amount, and we certainly have that now and have had that very much so over the past couple of months with the very steep slope of acceleration of cases that we've seen, is that the virus will continue to mutate and will mutate for its own selective advantage."
90
Anthony Fauci
Slightly Positive
00:33:16-00:33:39 (23 sec)
"So, if you have a lot of cases in Los Angeles and you have this mutant that you referred to, you can be almost certain that, as long as there's a lot of virus circulating in the community, there will be the evolution of mutants because that's what viruses do, particularly RNA viruses. And that's what I was referring to just a moment ago."
91
Anthony Fauci
Very Positive
00:33:39-00:34:02 (23 sec)
"You're giving the virus an opportunity to adapt. So, when people make it immune response against it, particularly in someone like -- that might be immunosuppressed where the virus stays in that person for a longer period of time, it gives the virus the chance to adapt to the forces, in this case the immune response, that's trying to get rid of it."
92
Anthony Fauci
Somewhat Positive

00:34:02-00:34:30 (28 sec)

"And that's where you get mutations. So, if the question you were asking, which is very relevant, is that what do we think is happening in other cities, I think what -- the underlying issues that are going on in California and Los Angeles very likely are taking place throughout the country, which is one of -- the reason why, as Dr. Walensky has said, that we are really ratcheting up our genomic surveillance capability and our ability to get that information in real time."
93
Andy Slavitt
Somewhat Positive

00:34:30-00:34:34 (4 sec)

"Dr. Walensky, anything you'd like to add, or is that --"
94
Rochelle Walensky
Slightly Negative

00:34:34-00:34:34 ( sec)

"-- No --"
95
Andy Slavitt
Slightly Positive

00:34:34-00:34:34 ( sec)

"-- Okay --"
96
Rochelle Walensky
Neutral

00:34:34-00:34:35 (1 sec)
"-- I think we got it."
97
Andy Slavitt
Very Positive

00:34:35-00:34:54 (19 sec)
"Okay. So, I -- I think what we're -- what we're saying collectively is let's not be quite such polite hosts to this virus. Let's turn the tide and do like other countries who do everything possible to shut out the growth of this virus and make sure it's not welcome. Well, thank you all for attending the briefing."
98
Andy Slavitt
Very Positive

00:34:54-00:35:14 (20 sec)
"I hope that this is useful to you. We intend to continue to do these every Monday, Wednesday, and Friday with the same group, and we will bring others on as well. So, if you have any feedback on how we can continue to share this information with the public, please let us know. Thank you and have a great weekend."
99